|
|
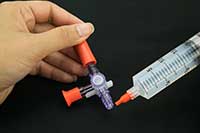
| White Paper: Find the Right Medical Adhesive |
Medical device engineers have to contend with a strict regulatory environment, and often have the toughest time striking the balance between end-use and manufacturability requirements. Master Bond’s latest white paper addresses five basic questions that will enable engineers to make a more educated choice when looking for the ideal medical adhesive for their application.

|
|
|
| Low Viscosity Epoxy Meets USP Class VI Specifications |
Ideal for encapsulation, bonding and sealing applications, Master Bond EP30Med offers excellent electrical insulation properties and exceptionally low shrinkage upon cure. It is a two component, room temperature curing biocompatible epoxy adhesive designed for high performance medical device applications. It features high bond strength, superior durability and outstanding chemical resistance. It also meets FDA requirements for indirect food contact as per FDA CFR 175.105.
For more information, request a technical data sheet on EP30Med |
|
|
| Biocompatible Epoxy for Medical Devices Resists Sterilization |
EP21LVMed is a two component, low viscosity epoxy resin system for high performance bonding, sealing, coating, encapsulating and casting. It produces high strength, durable bonds which hold up well to thermal cycling and resist many chemicals. Serviceable over the wide temperature range of -65°F to +250°F, EP21LVMed has a convenient one to one mix ratio.
For more information, request a technical data sheet on EP21LVMed |
|
|
| Medical Grade, Electrically Conductive Adhesive Paste |
EP3HTSMed is a one component, silver filled, electrically conductive, non-drip epoxy featuring high shear strength and a fast cure schedule at 250-300°F. This polymer compound fully meets USP Class VI requirements and has a low volume resistivity of <0.001 ohm-cm. EP3HTSMed produces durable bonds which are resistant to severe thermal cycling and many chemicals. It can be easily dispensed with syringe applicators.
For more information, request a technical data sheet on EP3HTSMed |
|
|
| High Viscosity, Optically Clear UV Curable Adhesive |
UV10TKMed is USP Class VI approved and meets ISO10993-5 for cytotoxicity requirements. It is used in the assembly of medical devices and is capable of withstanding repeated sterilizations, including radiation, ethylene oxide, chemical sterilants and especially autoclaving. This higher viscosity, one part system has exceptional optical clarity, sterling physical properties and a very high glass transition temperature (Tg) that exceeds 140°C.
For more information, request a technical data sheet on UV10TKMed |
|
|
| Addition Cured Silicone for High Performance Bonding and Sealing |
Formulated for medical device applications, MasterSil 153Med passes USP Class VI and ISO 10993-5 testing. This two component, addition cured system does not require exposure to air for complete cross-linking and has a convenient one to one mix ratio by weight. It will not outgas while curing. Other features include remarkable flexibility and high temperature resistance while maintaining very low exotherm over an exceptionally long working life.
For more information, request a technical data sheet on MasterSil 153Med |
|
|
| Biocompatible Silicone Passes USP Class VI Testing |
MasterSil 912Med is a single component silicone with a paste viscosity for bonding, sealing and coating medical devices. This non-corrosive, neutral cure compound complies with USP Class VI and ISO 10993-5 specifications. It resists many kinds of sterilization including gamma radiation, EtO and some types of liquid sterilants. The service temperature range extends from -75°F to +400°F. MasterSil 912Med withstands rigorous thermal cycling as well as thermal and mechanical shock.
For more information, request a technical data sheet on MasterSil 912Med |
|
|
|
Copyright  2015 by Master Bond Inc. All Rights Reserved. This content may not be reproduced in any way without the prior written permission of Master Bond Inc. 2015 by Master Bond Inc. All Rights Reserved. This content may not be reproduced in any way without the prior written permission of Master Bond Inc.
|
|
 View this email online
View this email online



 August 2015
August 2015