
The Application
University of Hawaii researchers, under the sponsorship of the Department of Energy, were looking to develop photoelectrochemical (PEC) systems to produce hydrogen directly from water using sunlight as the energy source. These PEC systems need to be highly efficient (offering solar-to-chemical conversion rates of 10-15%), potentially low cost and long operating lifetimes.
Key Parameters and Requirements
The researchers determined that the development of multijunction photoelectrodes comprised of a semiconductor, catalytic and protective thin films applied to low cost substrates like stainless steel was the key to achieving their goals. The photo electrodes were fabricated from amorphous silicon (a-Si) and copper-idium-gallium-diselenide (CIGS) and required the use of a highly transparent and stable polymer adhesive as a photoelectrode encapsulant.
The Results
Of several materials tested, the best results were obtained using the Mater Bond polymer system EP39-2, a transparent epoxy polymer, designed specifically for potting optoelectronic devices. This material was found to have excellent optical properties (see graph below) and good stability in potassium hydroxide (KOH). In addition, the material was compatible with traditional screenprinting techniques, which could be readily applied to the planar photoelectrode structure.
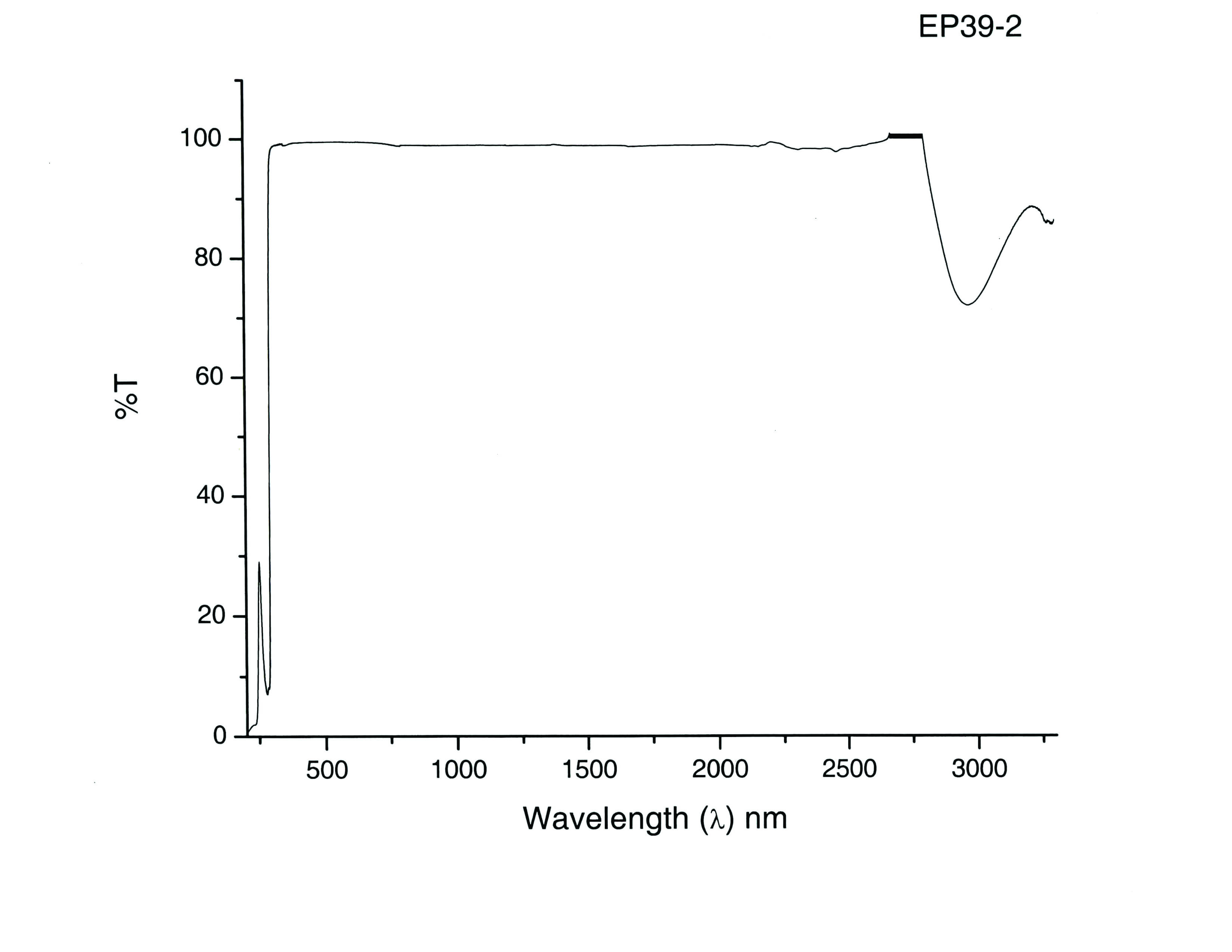
Transmission curve as tested by an independent lab
Sources
1 Miller, Eric, Richard Rocheleau. Photoelectrochemical Hydrogen Production, https://www1.eere.energy.gov/hydrogenandfuelcells/pdfs/30535x.pdf
